【CAS No.】4691-65-0
【E No.】E630
【Specification】FCCIV food grade
【Molecular Formula】C10H11N4Na2O8P
【Formula Weight】392.17
【Storage】Be stored in cool and dry place
【Chemical Properties】White to almost white powder. Soluble in water and formic acid, slightly soluble in ether, acetone and ethanol.
|
ABOUT DISODIUM 5'-RIBONUCLEOTIDE
VIADD CHEMICAL company is one professional DISODIUM 5'-RIBONUCLEOTIDE supplier, distibutor and exporter from China, welcome get price by email: sales@viaddchem.com
Disodium 5'-ribonucleotides, E number E635, is a flavor enhancer which is synergistic with glutamates in creating the taste of umami. It is a mixture of disodium inosinate (IMP) and disodium guanylate (GMP) and is often used where a food already contains natural glutamates (as in meat extract) or added monosodium glutamate (MSG). It is primarily used in flavored noodles, snack foods, chips, crackers, sauces and fast foods. It is produced by combining the sodium salts of the natural compounds guanylic acid (E626) and inosinic acid (E630).
A mixture composing of 98% monosodium glutamate and 2% E635 has four times the flavor enhancing power of monosodium glutamate (MSG) alone.[1]
I+G, is the flavor enhancers which are synergistic with glutamates in creating the taste of umami. It is a mixture of disodium insinate (IMP) and disodium guanylate (GMP) and is often used where a food already contains natural glutamates (as in meat extract) or added monosodium glutamate (MSG). It is primarily used in flavored noodles, snack foods, chips, crackers, sauces, and fast foods. It is produced by combining the sodium salts of guanylic acid and inosinic acid. A mixture of 98% monosodium glutamate and 2% disodium 5-ribonucleotides has four times the flavor enhancing power of mono-sodium glutamate alone.
Disodium 5'-ribonucleotides is an abbreviation for the combination between two condiments and by taking the initial English letters. It is made from mixing with respective 50% of IMP (DISODIUMINOSINE5’—MONOPHOSPHATE) and GMP (DISODIUM GUANOSINE5’—MONOPHOSPHATE)
【Basic Information】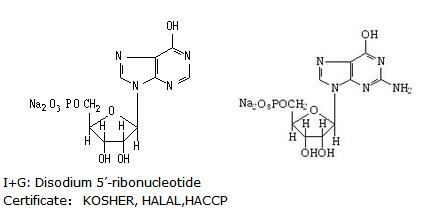
【Product Name】I+G/Disodium 5'-ribonucleotide
【CAS No.】4691-65-0
【E No.】E630
【Specification】FCCIV food grade
【Molecular Formula】C10H11N4Na2O8P
【Formula Weight】392.17
【Storage】Be stored in cool and dry place
【Chemical Properties】White to almost white powder. Soluble in water and formic acid, slightly soluble in ether, acetone and ethanol.

【Commercial Information】
【Packing】20kg or 25kg/bag/drum/carton box/requested
【Shelf Life】2 years
【Delivery】Within 15 days of order date
【Payment】T/T, D/P, L/C
【Origin】China
【Brand】VIADD
【Supplier】Shenyang Viadd Chemical Co.,Ltd.
【Price Email】sales@viaddchem.com
【Certificate】ISO,KOSHER,HALAL,GMP, MSDS, Material Data Sheet, Non GMO,Non Allergen
【Deliver Port】Tianjin/Qingdao
【Documents】Sales Contract, Commercial Invice, Packing List, Weight List, COA, , Cetificate of Origin, Healthey Certificate, FORM A, FORM E, Policy of Indusrance, B/L, Loading container photos, packing photos, shipping advice, etc
【I+G Characters】
1). Appearance: white crystal or powder;
2). Technological and quality parameters: all according to those of monosodium glutamate made in Japan;
3). Being used as delicious flavoring in diet, fast food, soup, soy, sauce and other kinds of snack.
【Product Feature】
1、Multiply effect in delicate flavor. When it is used with sodium glutamate, it can form multiplied improving for delicate flavor so as to reduce the product cost.
2、Enhance and improve food flavor; enhance the naturally delicious, strong, fragrant and sweet flavor.
3、It can make meat more delicious; when it mixed with monosodium glutamate and added into the meat, it can improve the meat original taste and enhance the meat delicate flavor, so that it can reduce meat consumption and reduce cost.
4、Restrain too salty, bitter, sour and other objectionable flavor and reduce foreign taste (amino acid taste or flour taste)
5、It is with good solubleness and stability in product.
【Specification】
|
Item |
Specification |
|
Transmittance (5%water solution) |
≥95.0 |
|
Assay(I+G), % |
97.0 to 102.0 |
|
IMP(mixed proportion), % |
48 to 52 |
|
GMP(mixed proportion), % |
48 to 52 |
|
Loss on drying, % |
25.0 |
|
Other related compounds of nucleic acid |
Not Detectable |
|
Heavy metals(Pb), % |
≤0.001 |
|
Arsenic(As), % |
≤0.0001 |
|
pH(5%water solution) |
7.0 to 8.5 |
|
Amino Acid |
Solution appear colorless |
|
NH4+(ammonium) |
Color of litmus paper unchanged |
【Application Field】
Disodium 5'-ribonucleotide is a new geneartonal nucleotide food taste agent. This product is prepared through the IMP (DISODIUMINOSINE5’—MONOPHOSPHATE) and GMP (DISODIUMGUANOSINE5’—MONOPHOSPHATE) are mixed with proportion of 1:1. It can be directly added in food and act taste improving effect. It is a kind of economic and best effective taste enhancer, and is also the main flavoring component in seasoning packet of instant noodles, seasoning, such as chicken extract, chicken powder, flavor enhancing soybean sauce and others. When mixing with sodium glutamate (monosodium glutamate), its dose is 2%-5% of monosodium glutamate, so that it also called “super monosodium glutamate”; in addition, it also have certain supportive therapeutic efficacy for motility hepatitis, chronic hepatitis, progressive muscular dystrophy and various kinds ophthalmic diseases.
I+G, can substitute flavors for natural ingredients. The flavor enhancement value of a mixture consisting of 3% of IMP & 97% of MSG is 3.8 times stronger than 100% MSG alone.
It can be used in various foods such as soup, canned foods, sauces, ketchup, juice, frozen foods, hamburger, processed cheese, dressing, processed meats, snacks, etc.
【Method of Applictaion】
Method of application: used as flavoring agent
Range of application: various kinds of food
Max application amount: appropriately added acording to the production demand.
I+G is usually used with monosodium glutamate to act collaborative flavor enhancing function; its dose is usually equal to the 0.5%~1.5% of monosodium glutamate. If added into soybean sauce, since the contained phosphatidase may decomposes I+G, should perform heat treatment to the soybean sauce before so as to realize enzyme denaturalixation.
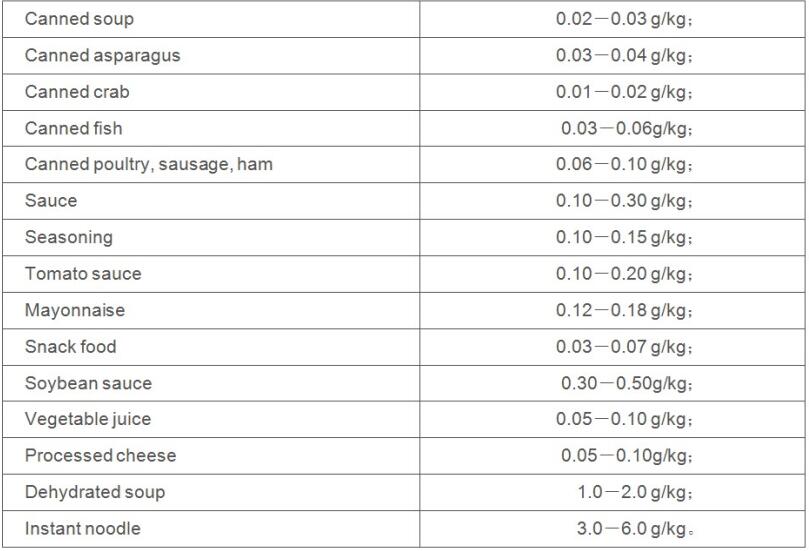
If the proportion between IMP and GMP is 1:1, its general dosage is as following:
【Side effects and safety】
Disodium 5'-ribonucleotides were first assessed in 1974 by the Joint FAO/WHO Expert Committee on Food Additives based on all available scientific literature. This assessment resulted in a new specification prepared and an "ADI Not Specified".[2] This essentially means that this additive shows no toxicology at any level and acceptable daily limits do not need to be set. The definition is as follows:
“This statement means that, on the basis of available data (chemical, biochemical, and toxicological), the total daily intake of the substance arising from its use or uses at levels necessary to achieve the desired effect and from its acceptable background in food, does not, in the opinion of the Committee, represent a hazard to health. For this reason, and for reasons stated in the individual evaluations, the establishment of an acceptable daily intake (ADI) expressed in mg per kg of body weight is not deemed necessary.”
In 1993 the Joint FAO/WHO Expert Committee on Food Additives considered several more studies on this food additive and retained the "ADI not specified" safety classification.